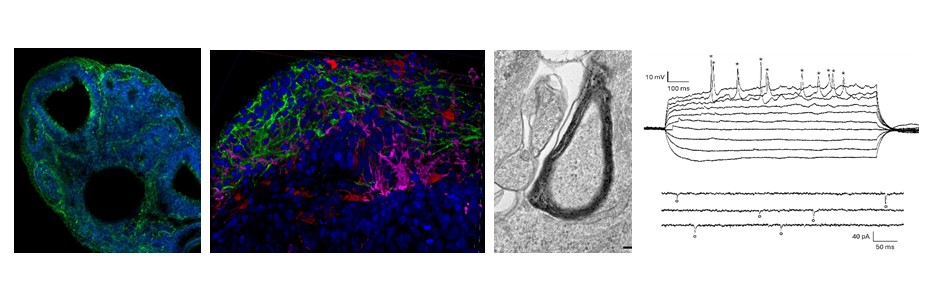
Our lab uses 2D and 3D human biomodels to investigate the mechanisms of brain development, aging and aging-related neurodegeneration, with particular emphasis on Parkinson’s diseases. The urgency to develop reliable human brain models derives from the historical difficulty to understand the patho-physiology of human brain and to observe on a dish the development of brain aging and neurodegenerative diseases. Our “Gold Standard” model is the iPSC-derived brain organoid. Brain organoids are generated from the donor’s stem cells to reproduce a 2-3 mm minibrain, maintaining the genetic and biochemical fingerprint of the donor. We apply the use of human biomodels in different running projects to study brain aging, Parkinson’s diseases and the rare disease CTNNB1, a neurodevelopmental disease classified as an autism spectrum disorder.
PROJECT 1. Brain aging dynamics in a novel human organoid model (organage).
The main objective of ORGANAGE is to identify the regulatory dynamics of human brain aging and the mechanisms of age-related neurodegeneration using a novel “aging-brain organoid”. In ORGANAGE we will use two different approaches to investigate the role of circulating peripheral factors and the direct effect of CNS cells in brain aging by transplanting differentiated organoids into the mouse brain.
PROJECT 2. Cellular and molecular effects of the SARS-CoV-2 viral proteins in human brain organoids.
In this project, we aim to study the development of SARS-CoV-2 infection in a brain with Parkinson’s disease (PD). We will treat human midbrain organoids derived from patients with PD and donors without PD with the recombinant spike protein of the SARS-CoV-2 virus and its receptor-binding domain. The interaction between the spike protein and the ACE2 protein in different cell types will be characterized to identify the virus’s cellular target. Additionally, we will analyze cell mortality and characterize glial activation, the neuroinflammatory profile, the integrity of dopaminergic cells, and electrophysiological activity. Finally, we will investigate the pathological effects of viral proteins in a Parkinson’s model in Drosophila. (in collaboration with S. Soukup and J.L. Zugaza).
PROJECT 3. Molecular basis and human biomodels of the CTNNB1 syndrome: therapeutic implications.
CTNNB1 syndrome is a neurodevelopmental disorder classified as a rare disease. The main clinical manifestations include language impairment, microcephaly, motor dysfunction, intellectual disability, behavioral problems, and autistic symptoms. The syndrome is associated with various mutations in the CTNNB1 gene, which encodes the β-catenin protein, a key effector in neurodevelopment. The aim of this project is to perform a structural and functional characterization of the β-catenin variants found in Spanish patients with CTNNB1 syndrome by generating different organoid models of the disease. (In collaboration with the CTNNB1 Spain Association and the IberoCTNNB1 Consortium).